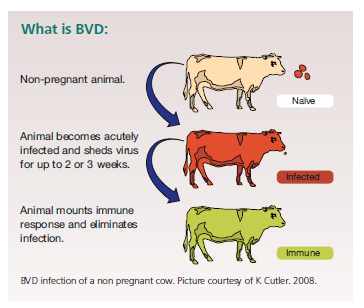
BVD eradication – Update and control measures – Bovilis
The incidence of BVD among Irish herds has declined dramatically since the introduction of the eradication programme in 2013. In 2013, there was 16,194
Read more about BVD eradication – Update and control measures – Bovilis





